The likelihood of remaining in response at 4 years was 29% (95% CI, 15%-45%) vs 0%, respectively. Median DOR with the triplet and chemotherapy alone was 17.5 months and 4.3 months, respectively. In the PD-L1 less than 1% population, the ORR was 31.1% (95% CI, 23%-40%) with the triplet vs 20.2% (95% CI, 14%-28%) with chemotherapy alone. The likelihood of remaining in response at 4 years was 25% (95% CI, 17%-33%) with the triplet vs 12% (95% CI, 6%-20%) with chemotherapy alone. 
Median duration of response (DOR) was 12.4 months with the triplet vs 5.6 months with chemotherapy alone. In the all-randomized population, the ORR was 38.0% (95% CI, 33%-43%) with the triplet vs 25.1% (95% CI, 21%-30%) with chemotherapy alone. OS by tumor histologic subtype served as an exploratory end point. 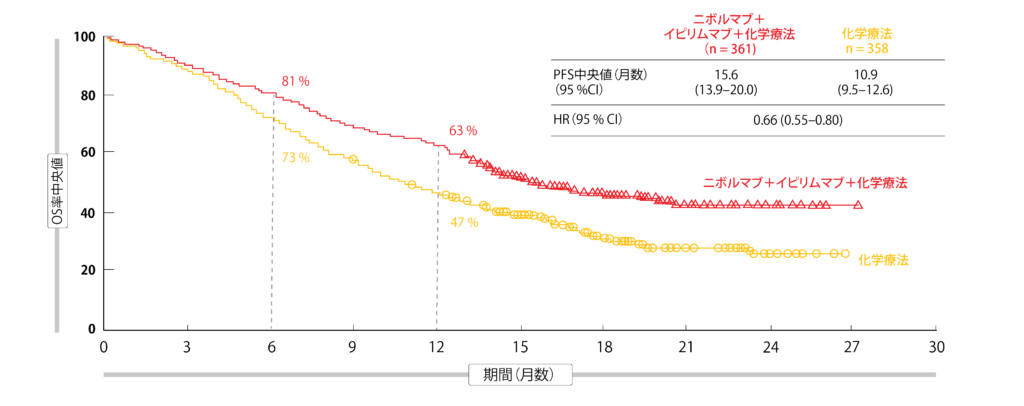
Secondary end points included progression-free survival (PFS), objective response rate (ORR), and efficacy by tumor PD-L1 expression. 
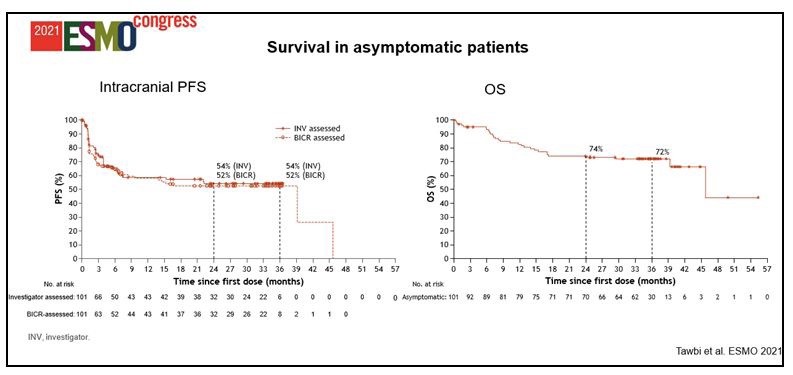
Treatment was continued until disease progression, unacceptable toxicity, or for 2 years for immunotherapy. A total of 719 patients were randomly assigned 1:1 to 360 mg of nivolumab every 3 weeks plus 1 mg/kg of ipilimumab every 6 weeks plus 2 cycles of chemotherapy given once every 3 weeks, or 4 cycles of chemotherapy alone once every 3 weeks with optional pemetrexed maintenance for patients with nonsquamous histology. The study enrolled patients with stage IV or recurrent NSCLC who had not received prior systemic therapy and had no sensitizing EGFR mutations or known ALK alterations. The triplet regimen is approved for the treatment of patients with metastatic NSCLC without EGFR or ALK alterations in the United States, European Union, and several other countries based on previously reported data from the pivotal trial. Bonner Chair in Lung Cancer Research, and director, James Thoracic Center, The Ohio State University Comprehensive Cancer Center–James in Columbus, and co-authors, wrote in a poster of the data. “With a 4-year minimum follow-up, patients treated with nivolumab plus ipilimumab and chemotherapy continued to derive long-term, durable OS benefit vs chemotherapy alone, regardless or tumor PD-L1 expression or histology,” lead study author David P. In the nonsquamous population, median OS was 17.8 months with the triplet (n = 246) vs 12.0 months with chemotherapy alone (n = 246 HR, 0.80 95% CI, 0.66-0.97). In the PD-L1–positive population (PD-L1 ≥1%), median OS was 15.8 months with the triplet (n = 204) vs 10.9 months with chemotherapy alone (n = 204 HR, 0.74 95% CI, 0.60-0.92) four-year OS rates were 21% vs 16%, respectively.īroken down by histology, median OS was 14.5 months with the triplet (n = 115) vs 9.1 months with chemotherapy alone (n = 112) in the squamous population (HR, 0.64 95% CI, 0.48-0.84). In the PD-L1 less than 1% population, median OS was 17.7 months with the triplet (n = 135) vs 9.8 months with chemotherapy alone (n = 129), with 4-year OS rates of 23% vs 13%, respectively (HR, 0.66 95% CI, 0.50-0.86). In all patients who were randomly assigned, median overall survival (OS) was 15.8 months with the triplet (n = 361) vs 11.0 months with chemotherapy alone (n = 358 HR, 0.74 95% CI, 0.63-0.87) four-year OS rates were 21% and 16%, respectively. In the phase 3 CheckMate 9LA trial (NCT03215706), patients with advanced non–small cell lung cancer (NSCLC), particularly those with PD-L1–negative and squamous disease, derived durable benefit from first-line treatment with nivolumab (Opdivo) plus ipilimumab (Yervoy), results presented at the 2023 ASCO Annual Meeting show. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
